- With good calenderability, abrasive resistance, corrosion resistance, castability, and room temperature mechanical properties, zinc can be made into various alloys with many other metals. Mainly in the form of galvanization, zinc-based alloys and zinc oxide, it has applications in the automobile, construction and shipbuilding industries, light industry, machinery, household electrical appliances, batteries and other industries. Currently, its consumption among non-ferrous metals is second only to aluminum and copper.
 As in moist air, a protective layer is easily produced on the surface of zinc, which prevents further atmospheric corrosion. zinc is widely used in the galvanization industry. Galvanization is primarily used in steel and surface coatings on steel structures (eg. galvanized sheet) for automobiles, construction, shipbuilding, light and some other industries. Examples include: coatings containing zinc powder; zinc spelter, used in connections (eg. steel components connecting ships, bridges and offshore oil and gas derricks), roofing made of galvanized iron sheet; and steel strip hot-dip galvanization.
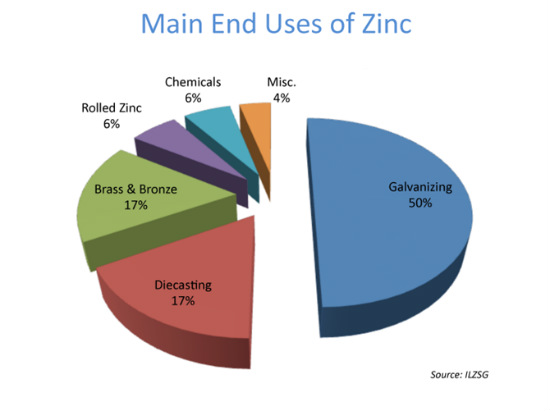
As in moist air, a protective layer is easily produced on the surface of zinc, which prevents further atmospheric corrosion. zinc is widely used in the galvanization industry. Galvanization is primarily used in steel and surface coatings on steel structures (eg. galvanized sheet) for automobiles, construction, shipbuilding, light and some other industries. Examples include: coatings containing zinc powder; zinc spelter, used in connections (eg. steel components connecting ships, bridges and offshore oil and gas derricks), roofing made of galvanized iron sheet; and steel strip hot-dip galvanization. - At present, galvanization accounts for half of all zinc consumption.
 Though the intensity and hardness of zinc are not good, it has valuable mechanical properties, and when combined with aluminum and copper to form zinc alloys, its intensity and hardness improve greatly. In particular, the comprehensive mechanical properties of zinc-copper-titanium alloy are close, if not equal to, those of aluminum alloy, brass or gray cast iron; moreover, creep resistance is improved substantially. Therefore, zinc alloys are widely used in the production of many components and die-casting fittings in automobile manufacturing and the mechanical industry, thanks to its superior superplasticity. In addition, printing zinc plates, powder corrosion photographic plates and offset printing plates can be made of zinc plate, consuming less lead, cadmium and other elements.
Though the intensity and hardness of zinc are not good, it has valuable mechanical properties, and when combined with aluminum and copper to form zinc alloys, its intensity and hardness improve greatly. In particular, the comprehensive mechanical properties of zinc-copper-titanium alloy are close, if not equal to, those of aluminum alloy, brass or gray cast iron; moreover, creep resistance is improved substantially. Therefore, zinc alloys are widely used in the production of many components and die-casting fittings in automobile manufacturing and the mechanical industry, thanks to its superior superplasticity. In addition, printing zinc plates, powder corrosion photographic plates and offset printing plates can be made of zinc plate, consuming less lead, cadmium and other elements.- Currently, zinc alloy makes up around 20% of zinc applications. However, in western countries, they have realized that zinc alloy can be directly used as the covering material for roofs, extending the service life from 5-10 years to 120-140 years- what’s more, the material can be recycled. Thus, in the long run, when it comes to zinc consumption, the ratio of galvanization will decrease, while that of zinc alloy will gradually increase.
- Zinc can be used to make batteries, such as zinc-manganese batteries and zinc air batteries.
- In addition, zinc has good electromagnetic field resistance properties. In the case of radio frequency interference, zinc plate is a very effective shielding material.
- And as zinc is non-magnetic, it is suitable for making components and covers of instruments and meters.
- Since zinc produces no sparks, either alone or in collision with other metals, it is suitable for making explosion-proof equipment.
- Zinc fertilizer (eg. zinc sulfate and zinc chloride) can promote plant cell respiration and carbohydrate metabolism.
- Zinc powder, lithopone and zinc chrome can be made into pigments.
- Zinc oxide can also be used in the pharmaceutical, rubber, paint and other industries.
- Main end uses of zinc is as follows:
-
About us
Contact us
Make a suggestion
- Metalpedia is a non-profit website, aiming to broaden metal knowledge and provide extensive reference database to users. It provides users reliable information and knowledge to the greatest extent. If there is any copyright violation, please notify us through our contact details to delete such infringement content promptly.
 As in moist air, a protective layer is easily produced on the surface of zinc, which prevents further atmospheric corrosion. zinc is widely used in the galvanization industry. Galvanization is primarily used in steel and surface coatings on steel structures (eg. galvanized sheet) for automobiles, construction, shipbuilding, light and some other industries. Examples include: coatings containing zinc powder; zinc spelter, used in connections (eg. steel components connecting ships, bridges and offshore oil and gas derricks), roofing made of galvanized iron sheet; and steel strip hot-dip galvanization.
As in moist air, a protective layer is easily produced on the surface of zinc, which prevents further atmospheric corrosion. zinc is widely used in the galvanization industry. Galvanization is primarily used in steel and surface coatings on steel structures (eg. galvanized sheet) for automobiles, construction, shipbuilding, light and some other industries. Examples include: coatings containing zinc powder; zinc spelter, used in connections (eg. steel components connecting ships, bridges and offshore oil and gas derricks), roofing made of galvanized iron sheet; and steel strip hot-dip galvanization.  Though the intensity and hardness of zinc are not good, it has valuable mechanical properties, and when combined with aluminum and copper to form zinc alloys, its intensity and hardness improve greatly. In particular, the comprehensive mechanical properties of zinc-copper-titanium alloy are close, if not equal to, those of aluminum alloy, brass or gray cast iron; moreover, creep resistance is improved substantially. Therefore, zinc alloys are widely used in the production of many components and die-casting fittings in automobile manufacturing and the mechanical industry, thanks to its superior superplasticity. In addition, printing zinc plates, powder corrosion photographic plates and offset printing plates can be made of zinc plate, consuming less lead, cadmium and other elements.
Though the intensity and hardness of zinc are not good, it has valuable mechanical properties, and when combined with aluminum and copper to form zinc alloys, its intensity and hardness improve greatly. In particular, the comprehensive mechanical properties of zinc-copper-titanium alloy are close, if not equal to, those of aluminum alloy, brass or gray cast iron; moreover, creep resistance is improved substantially. Therefore, zinc alloys are widely used in the production of many components and die-casting fittings in automobile manufacturing and the mechanical industry, thanks to its superior superplasticity. In addition, printing zinc plates, powder corrosion photographic plates and offset printing plates can be made of zinc plate, consuming less lead, cadmium and other elements.
